RESEARCH BRIEFING
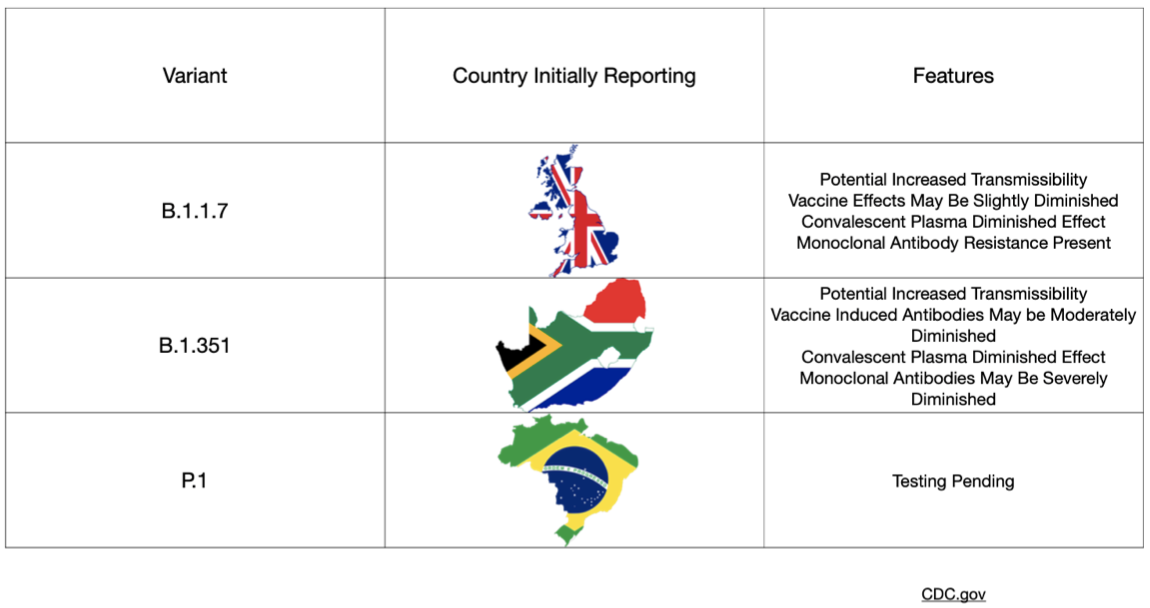
The covid-19 variants have dominated the news lately with significant focus paid to the UK (B.1.1.7), Brazilian (P.1), and South African (B.1.351) mutations. Concerns have been raised about increased infectiousness and the potential for increased mortality associated with these variant strains. In particular, many worry that the South African strain has even higher rates of transmission and imposes a higher viral burden than the UK version. Along with these concerns, questions have arisen regarding the effectiveness of our current vaccines and of natural immunity from previous infection with SARS-CoV-2. A recent group of preprints sought to address these questions.
The first preprint from researchers in the United States investigated the effectiveness of the mRNA-1273 vaccine (Moderna) against the aforementioned global variants. The Moderna vaccine previously demonstrated ~94 percent efficacy in Phase 3 data published last month and reviewed here at Brief19. A key feature of these variants is mutation of the spike proteins (the proteins studding the outer shell of the SARS-CoV-2 virus) with more extensive changes seen in the UK variant. Changes to parts of the protein called the "receptor binding domain" and "N-terminal domain" potentially have the ability to affect the binding of monoclonal antibody treatments, and crucially, possibly vaccine-induced antibodies too. This paper presents results from non-human primates and human subjects who received the Moderna vaccine. Blood samples from subjects were tested against multiple strains of the virus including the UK and South African mutations. Importantly and somewhat reassuringly the vaccine in these tests shows activity against the mutations—though some reduced effects were seen when exposed to the South African mutation in particular.
A second preprint investigated whether antibodies generated after a natural course of infection would provide resistance to the South African variant. In this very small study, convalescent plasma collected from six adult patients who were hospitalized with covid-19 in South Africa were then tested against the mutated strain. These results found that the natural antibodies demonstrated significant variation between the six test subjects, which raises concern that previous infection may not provide enough protection from variant exposure in all cases. These findings also imply that vaccines targeting the spike protein could only mount a weak response. The clinical implications of this are not understood.
A third preprint also addressed this increased resistance of the UK and South African variants to antibody neutralization. This study queried blood samples from those who had already received vaccination and those with naturally occurring antibodies generated after an infection (convalescent plasma), as well as efficacy of monoclonal (lab manufactured) antibodies. A total of 12 monoclonal antibodies were assessed along with convalescent plasma from 20 patients, plus samples from 12 people who enrolled in the Moderna Phase 1 study. Similar to the aforementioned preprints, the findings were concerning—the UK and South African variants demonstrated resistance to monoclonal antibodies, reduced efficacy of convalescent plasma, and a loss in vaccine activity ranging from 2 to 8.6 times less effective in the UK and South African variants, respectively. Again, the degree to which this would have a clinical impact is not known.
Moderna released a statement on January 25 hoping to assuage such fears, stating that their vaccine showed robust activity against these new strains. The company acknowledged the reduced effect, but still believe that titer levels generated by the vaccine should be protective. Moderna also suggest that the waning efficacy could be bolstered by a booster vaccine in the future.
In summary, these findings raise concern about potential for new infections, even in those previously infected or vaccinated. The usual scrutiny must be applied to these studies given they are preprints and all contain very small subject numbers. We must not let our guard down and continue safe practices of mask wearing and social distancing. Given the rapid appearance of mutations, the likelihood of annual vaccinations could be inevitable. The single best way to limit the emergence of even more worrying mutations remains to limit the spread of SARS-CoV-2. The fewer replication cycles it undergoes, the fewer mutations will occur.
POLICY BRIEFING
Yesterday the New York Attorney General released a report claiming that the state's health department had underreported covid-19 associated deaths by more than fifty percent. Specifically, an investigation was launched to evaluate the discrepancy in deaths reported by nursing homes and those presented by state health officials and found that only deaths that occurred in hospitals were being appropriately reported. While this discovery is surely a political blow to a state that has touted the robustness of its pandemic response and ability to contain the spread, it serves to highlight a more endemic problem: proper data management.
In June, the National Institutes of Health (NIH) launched a national database to join disparate hospital data sets in order to analyze potential patterns within the course SARS-CoV-2 infection, but this was voluntary and its intent was to guide policy around therapeutics. Since August, the US Centers for Disease Control and Prevention (CDC) and the broader US Department of Health and Human Services (HHS) have waged a turf war over data ownership. In an effort to create stability, the Centers for Medicare and Medicaid Services (CMS) eventually mandated that hospitals report directly to the HHS in order to receive Medicare reimbursement, effectively cutting the CDC out of the process. This created a shifting administrative burden based on changing data requirements and led to lapses in data reporting. While the move did standardize the information flowing from hospitals, it did so at the expense of the nation's premiere epidemiological entity and did not address the reporting from state and local health entities.
As PPE, disaster relief, and funds are all tied in some way to severity of regional disease burden, there needs to be a national repository with standard definitions that permit adequate and more importantly, accurate, understanding of the state of the pandemic in various regions around the country. Various.